Finding the Ration of Moles of Reactants
in a Chemical Reaction
PURPOSE
The purpose of this lab is to utilize the method of continuous variation in order to find the mole ratio in a chemical reaction.
TABLE
|
Sodium thiosulfate
(Bleach) in mL |
Sodium hypochlorite
in mL |
Initial temperature
(°C) |
Post-reaction Temperature
(°C) |
Change in Temperature
( °C) |
|
50.0 mL
45.0 mL 30.0 mL 20.0 mL 5.0 mL 0 mL |
0 mL
5.0 mL 20.0 mL 30.0 mL 45.0 mL 50.0 mL |
22°C
22°C 22°C 22°C 22°C 22 °C |
22°C
25.5 °C 36.0°C 32.5°C 24.5°C 22°C |
_____
3.5°C 14.0°C 10.5°C 2.5°C _____ |
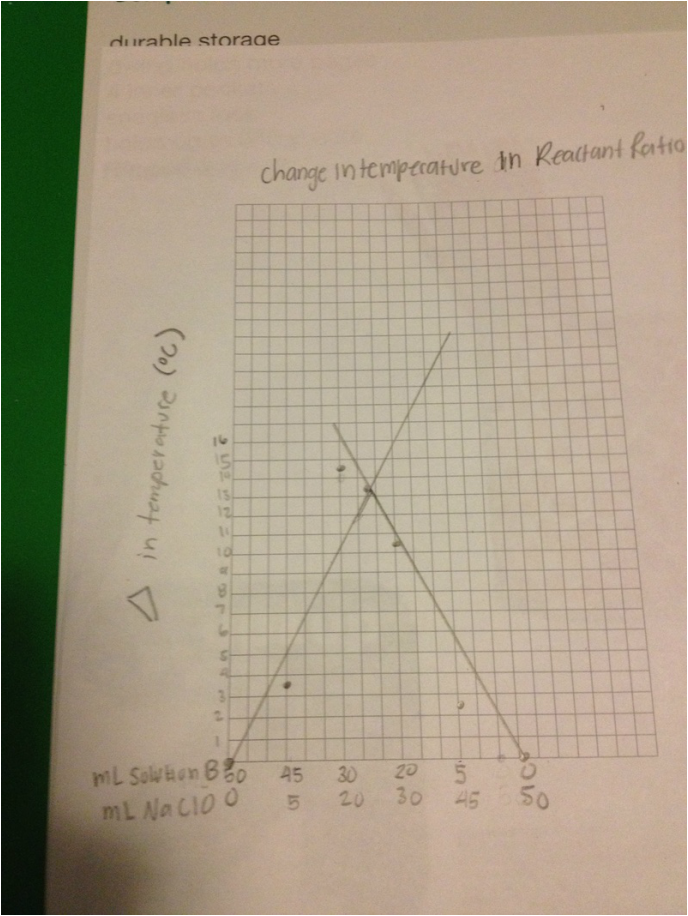
GRAPH
CONCLUSIONS
To conclude this experiment, it is safe to say that the usage of the different amounts of Solution B and NaClO, when mixed, were able to change temperatures. When mixing a certain amount of Solution B with a certain amount of NaClO and then reversing the amounts, there was a clear difference with the two products. For example, when mixing 45 mL of NaClO with 5 mL of Solution B, the temperature of the product was 24.5°C. When reversing the amounts, the temperature of the product became 25.5°C. When graphing the points, the intersection showed that at 28 mL for Solution B and 23 mL at NaClO, the temperature change that is produced is 13.4°C. The ratio of the two reactants was 0.0115 moles of NaClO and 0.014 moles of Solution B. This resulted in 6 moles of Solution B and 5 moles of NaClO after evening out the coefficients.
DISCUSSION OF THEORY
Using the method of continuous variation, the molar ratios were able to be found. This method is possible because of the consistency of the volume of both of the reactants. The mixing of the two solutions that contained different molar ratios was able to help expose the dependent variable, which was the change in temperature. Using this information would help observers determine the ratio of moles of reactants in a chemical reaction. At the greatest ratio, there would be the greatest temperature gain. This should at the least help determine the accurate molar ratio.
ANALYSIS QUESTIONS
1) It is essential to keep a constant volume of reactants because it is necessary to keep the amount of the solutions and the temperature proportional. Without a constant volume, there would not be anything to compare the results with. If there was not a constant volume, comparing the temperature increase would not mean anything. The consistency of the volume can be used to identify the highest and lowest temperature change.
2) The term "limiting reagent" can be defined as the reactant that is completely consumed and limits the amount of product that can be formed. The limiting reactant determines how much product is formed. After the limiting reagent is found, the remainder would be called the excess.
3)The temperature limits the precision of the data more than the volume. The temperature was measured with a thermometer. This could be seen as a problem because of how inaccurate the thermometer can be. It is hard read the measurements of the thermometer because the increments in between the ticks cannot be measured accurately. There can be so many errors that could happen with a misread calculation. Since the volume was constant, the measurement should have been the same throughout the experiment. Because of the consistency, the precision in relations to the volume should be fairly close.
4) Along the upward sloping line on the graph, the limiting reagent is NaClO. Along the the downward sloping line, the limiting reagent is Solution B.
5) Other than temperature change, some physical properties that can be used for the method of continuous variations could be color intensity of a reactant or product, the mass of a precipitate that forms, or the volume of a gas evolved.
6) It is more accurate to use the point of intersection of the two lines to find the mole ratio, rather than the ratio associated with the greatest temperature change because the point of intersection shows the complete maximum temperature change. The ratio of the greatest temperature change would not show the many factors in the reactants like volume.
2) The term "limiting reagent" can be defined as the reactant that is completely consumed and limits the amount of product that can be formed. The limiting reactant determines how much product is formed. After the limiting reagent is found, the remainder would be called the excess.
3)The temperature limits the precision of the data more than the volume. The temperature was measured with a thermometer. This could be seen as a problem because of how inaccurate the thermometer can be. It is hard read the measurements of the thermometer because the increments in between the ticks cannot be measured accurately. There can be so many errors that could happen with a misread calculation. Since the volume was constant, the measurement should have been the same throughout the experiment. Because of the consistency, the precision in relations to the volume should be fairly close.
4) Along the upward sloping line on the graph, the limiting reagent is NaClO. Along the the downward sloping line, the limiting reagent is Solution B.
5) Other than temperature change, some physical properties that can be used for the method of continuous variations could be color intensity of a reactant or product, the mass of a precipitate that forms, or the volume of a gas evolved.
6) It is more accurate to use the point of intersection of the two lines to find the mole ratio, rather than the ratio associated with the greatest temperature change because the point of intersection shows the complete maximum temperature change. The ratio of the greatest temperature change would not show the many factors in the reactants like volume.